Understanding The Formula Of Hypochlorous Acid: A Complete Guide
What is the formula of hypochlorous acid, and why does it matter? This question often arises in chemistry studies, water treatment discussions, and even healthcare applications. Hypochlorous acid, a weak acid with powerful oxidizing properties, plays a vital role in disinfection, cleaning, and biological processes. Its chemical formula, HOCl, is simple yet significant, as it determines how the compound behaves in different environments. Whether you're a student, a professional, or simply curious, understanding this formula can unlock insights into its uses and benefits.
Hypochlorous acid is widely recognized for its effectiveness in killing bacteria, viruses, and other pathogens. It is a naturally occurring compound produced by the human immune system to fight infections. In industrial applications, it is commonly used in water purification, sanitization, and even wound care. Despite its widespread use, many people remain unaware of its chemical composition and how it works. By exploring the formula HOCl, we can better appreciate its role in various fields and its importance in maintaining hygiene and health.
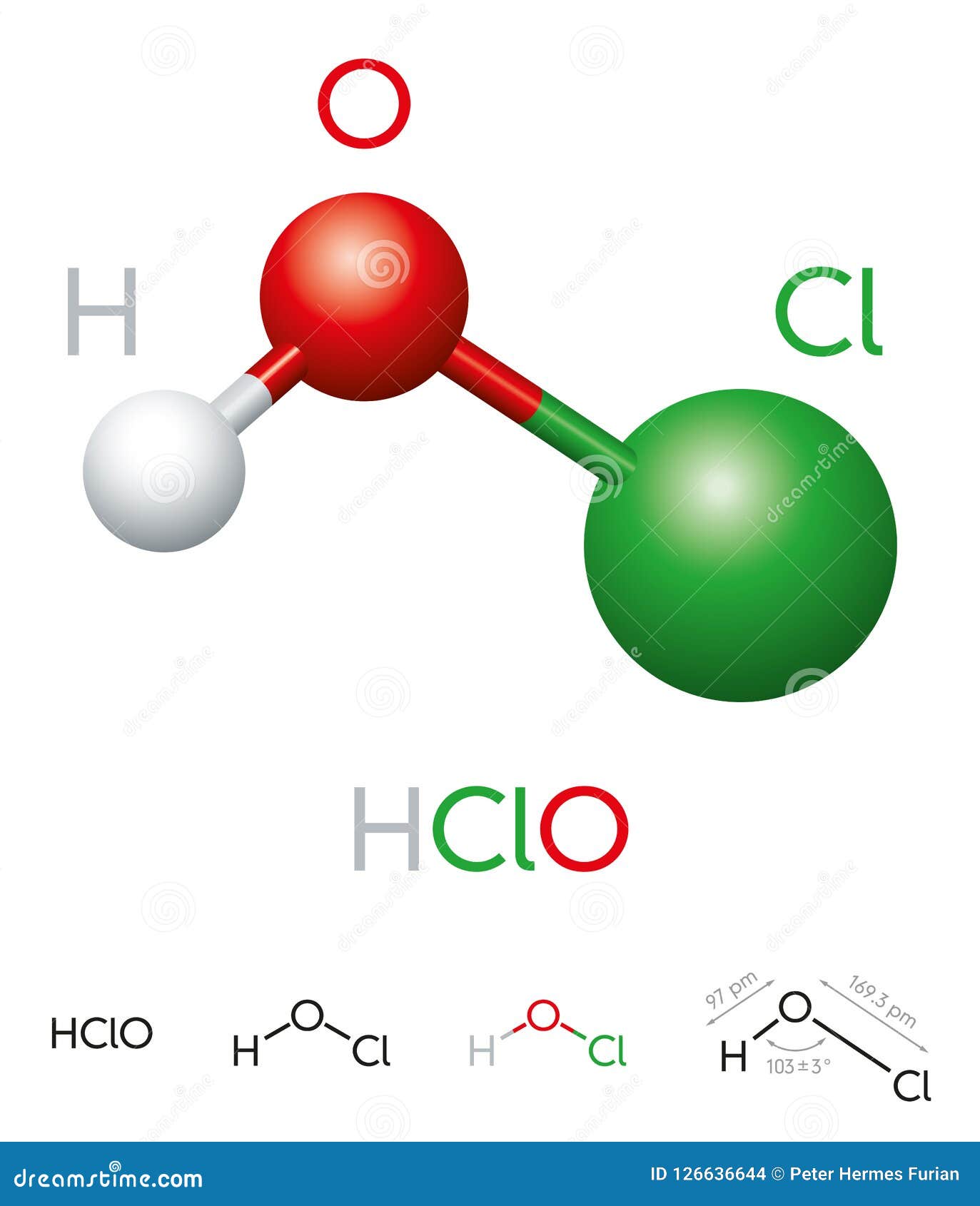
The chemical structure of hypochlorous acid consists of one hydrogen (H) atom, one oxygen (O) atom, and one chlorine (Cl) atom. This simple arrangement gives the compound its unique properties, including its ability to act as a powerful disinfectant. Understanding the formula of hypochlorous acid is not just about memorizing HOCl; it’s about grasping its implications in real-world applications. From swimming pools to medical facilities, this compound proves its worth time and again. Let’s dive deeper into the topic to uncover more about its formula, properties, and uses.
Read also:Exploring The Hottest Mms Viral Video News 2024
Table of Contents
- What is Hypochlorous Acid?
- What is the Formula of Hypochlorous Acid?
- How is Hypochlorous Acid Used?
- Is Hypochlorous Acid Safe for Human Use?
- What Are the Properties of Hypochlorous Acid?
- How Does Hypochlorous Acid Work?
- Can Hypochlorous Acid Be Used in Water Purification?
- What Are the Benefits of Hypochlorous Acid?
- How is Hypochlorous Acid Produced?
- Frequently Asked Questions
What is Hypochlorous Acid?
Hypochlorous acid is a weak acid that exists in equilibrium with its conjugate base, hypochlorite ion (OCl⁻). It is a naturally occurring compound that plays a crucial role in the immune system of humans and animals. When white blood cells encounter pathogens, they produce hypochlorous acid to neutralize them. This acid is also synthesized for industrial and commercial purposes, where it serves as a powerful disinfectant and sanitizer.
One of the key reasons hypochlorous acid is so effective is its ability to oxidize the cell walls of microorganisms, leading to their destruction. This makes it a preferred choice for applications where safety and efficacy are paramount. Unlike harsh chemicals, hypochlorous acid is biodegradable and non-toxic, making it suitable for a wide range of uses.
What is the Formula of Hypochlorous Acid?
The chemical formula of hypochlorous acid is HOCl. This formula indicates that the compound consists of one hydrogen atom, one oxygen atom, and one chlorine atom. The arrangement of these atoms gives hypochlorous acid its unique properties, including its ability to act as a strong oxidizing agent. Understanding this formula is essential for anyone studying chemistry or working in fields where hypochlorous acid is used.
What is the formula of hypochlorous acid? It’s HOCl, a simple yet powerful combination that has far-reaching implications. This compound exists in equilibrium with hypochlorite ions, depending on the pH of the solution. At lower pH levels, hypochlorous acid dominates, while at higher pH levels, hypochlorite ions become more prevalent. This balance is critical in determining the effectiveness of the acid in various applications.
How is Hypochlorous Acid Used?
Hypochlorous acid is used in a variety of industries due to its disinfectant properties. Some common applications include:
- Water purification
- Sanitization of surfaces
- Medical and healthcare settings
- Food processing and preservation
- Swimming pool maintenance
In water purification, hypochlorous acid is added to eliminate harmful microorganisms. Its ability to oxidize pathogens makes it an ideal choice for ensuring safe drinking water. In healthcare, it is used for wound cleaning and sterilization of medical equipment. Its non-toxic nature makes it safe for use even on sensitive skin.
Read also:Matt Czuchry Is He Married And What You Need To Know
Is Hypochlorous Acid Safe for Human Use?
Yes, hypochlorous acid is generally considered safe for human use when used appropriately. It is non-toxic, non-irritating, and biodegradable, making it a preferred alternative to harsh chemicals like bleach. However, it is important to follow guidelines and use it in the recommended concentrations to avoid any potential risks.
What Are the Properties of Hypochlorous Acid?
Hypochlorous acid possesses several unique properties that make it highly effective as a disinfectant:
- Powerful oxidizing agent
- Effective at low concentrations
- Biodegradable and environmentally friendly
- Non-toxic and non-irritating
- Works quickly to kill pathogens
How Does Hypochlorous Acid Work?
Hypochlorous acid works by oxidizing the cell walls of microorganisms, leading to their destruction. This process disrupts the structural integrity of bacteria, viruses, and fungi, rendering them unable to survive. Its effectiveness is influenced by factors such as pH, concentration, and contact time.
Can Hypochlorous Acid Be Used in Water Purification?
Yes, hypochlorous acid is widely used in water purification processes. Its ability to kill pathogens quickly and safely makes it an ideal choice for ensuring clean and safe drinking water. It is also used in swimming pools to maintain hygiene and prevent the spread of waterborne diseases.
What Are the Benefits of Hypochlorous Acid?
Hypochlorous acid offers numerous benefits, including:
- Highly effective disinfection
- Environmentally friendly
- Safe for human use
- Cost-effective
- Versatile applications
How is Hypochlorous Acid Produced?
Hypochlorous acid is produced by dissolving chlorine gas in water or through the electrolysis of saltwater. The process involves creating a solution that contains hypochlorous acid and hypochlorite ions. The pH of the solution determines the ratio of these two forms.
Frequently Asked Questions
What is the formula of hypochlorous acid?
The formula of hypochlorous acid is HOCl. It consists of one hydrogen atom, one oxygen atom, and one chlorine atom.
Is hypochlorous acid safe for skin?
Yes, hypochlorous acid is safe for skin when used in appropriate concentrations. It is non-irritating and biodegradable, making it suitable for sensitive skin.
How long does hypochlorous acid remain effective?
Hypochlorous acid remains effective for a limited time, as it is unstable and decomposes quickly. Proper storage and usage guidelines should be followed to maximize its effectiveness.
Can hypochlorous acid be used in food processing?
Yes, hypochlorous acid is commonly used in food processing for sanitization purposes. It is safe, effective, and leaves no harmful residues.
Understanding the formula of hypochlorous acid and its applications can provide valuable insights into its role in various industries. From water purification to healthcare, this compound continues to prove its worth as a safe and effective disinfectant.
Miscarriage By Week: A Comprehensive Guide To Understanding And Coping With Pregnancy Loss
Discovering The Life And Legacy Of Jeana Ellen Keough
Is Sara Driver Related To Adam Driver? Unveiling The Truth Behind The Names
HClO Hypochlorous Acid Molecule Model and Chemical Formula Stock Vector

MJ6001 Hypochlorous Acid bactericide RAYMOND