Understanding The Chemical Formula For Hypochlorous Acid: A Comprehensive Guide
Hypochlorous acid is a powerful yet gentle compound with a wide range of applications, from disinfection to wound care. Its chemical formula, HOCl, is simple yet highly effective in its role as an oxidizing agent. This weak acid is widely recognized for its ability to kill bacteria, viruses, and fungi without causing harm to human tissues, making it an essential component in various industries. Whether you're a student, a researcher, or someone curious about chemistry, understanding the chemical formula for hypochlorous acid can open doors to its diverse uses and benefits.
Hypochlorous acid is naturally produced in the human body by white blood cells to fight infections. It is also synthesized in laboratories for commercial purposes, such as water treatment, sanitization, and even skincare. The chemical formula for hypochlorous acid plays a crucial role in its functionality, as the balance of hydrogen, oxygen, and chlorine atoms determines its potency and stability. This article dives deep into the science behind HOCl, its properties, and its applications, ensuring you gain a thorough understanding of this fascinating molecule.
From its discovery to modern-day uses, hypochlorous acid has been a cornerstone in the field of chemistry and health sciences. Its ability to act as a disinfectant without leaving harmful residues has made it a preferred choice for eco-friendly solutions. In this guide, we will explore the chemical formula for hypochlorous acid in detail, answer common questions, and provide insights into its significance in various fields. By the end of this article, you’ll have a clear understanding of why this compound is so vital.
Read also:Exploring The Legacy Of James Caan And Scott Caan A Fatherson Journey In Hollywood
Table of Contents
- What is Hypochlorous Acid?
- The Chemical Formula for Hypochlorous Acid
- How Does Hypochlorous Acid Work?
- What Are the Applications of Hypochlorous Acid?
- Is Hypochlorous Acid Safe to Use?
- How is Hypochlorous Acid Produced?
- What Are the Chemical Properties of Hypochlorous Acid?
- What Are the Benefits of Hypochlorous Acid?
- Common Misconceptions About Hypochlorous Acid
- Frequently Asked Questions About Hypochlorous Acid
What is Hypochlorous Acid?
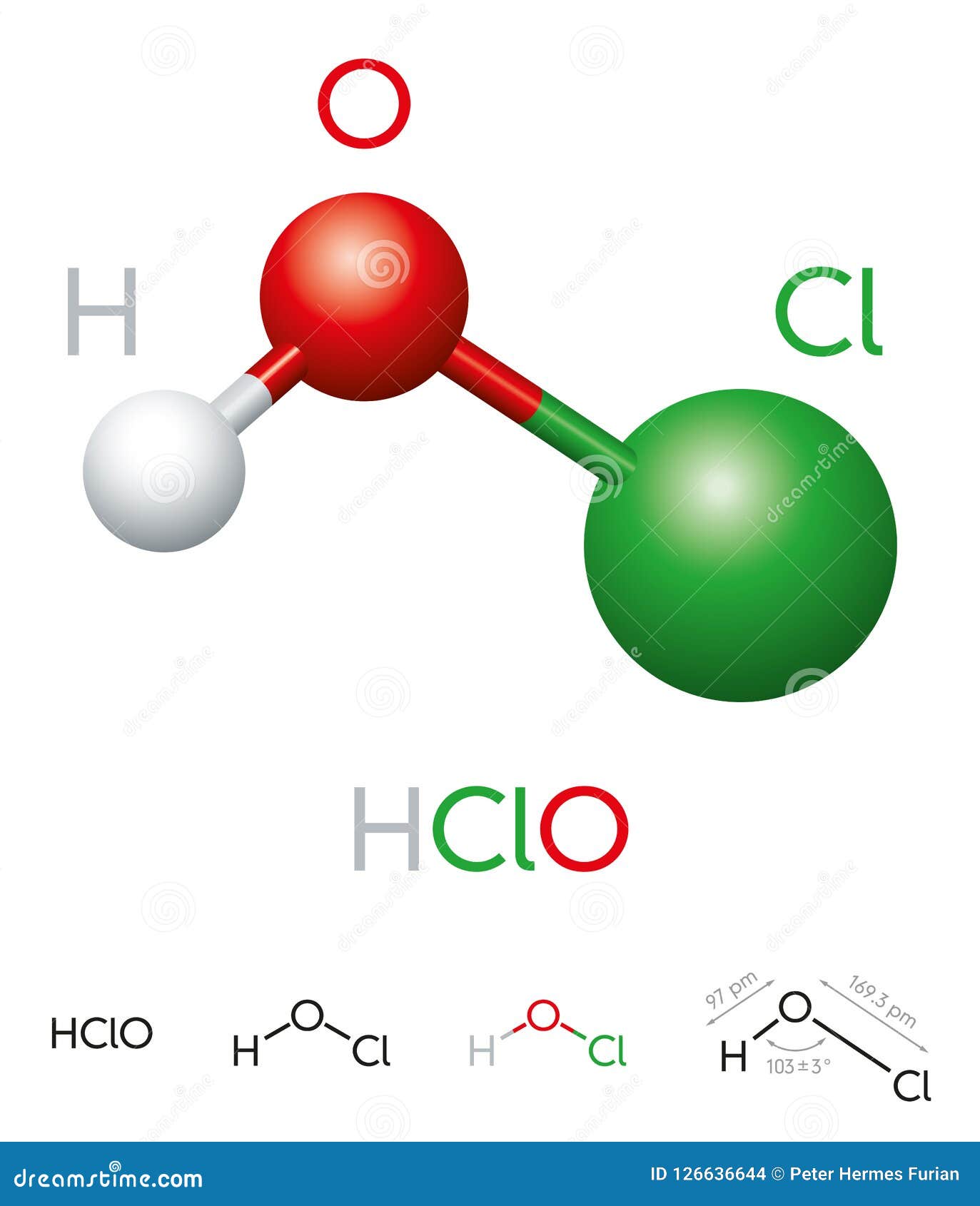
Hypochlorous acid is a weak acid that exists in solution, typically formed when chlorine dissolves in water. It is a naturally occurring substance in the human immune system, where it plays a critical role in defending the body against pathogens. Hypochlorous acid is highly reactive, which makes it effective in breaking down the cell walls of microorganisms. Its chemical formula, HOCl, indicates that it consists of one hydrogen atom, one oxygen atom, and one chlorine atom.
This compound is widely used in industries such as healthcare, agriculture, and water treatment due to its antimicrobial properties. Unlike other disinfectants, hypochlorous acid is non-toxic and biodegradable, making it an environmentally friendly option. Its ability to neutralize harmful microorganisms without leaving harmful residues has made it a preferred choice for sanitization and disinfection.
The Chemical Formula for Hypochlorous Acid
The chemical formula for hypochlorous acid is HOCl. This simple yet effective formula represents a molecule that is highly effective in killing pathogens. The hydrogen atom (H) is bonded to the oxygen atom (O), which is further bonded to the chlorine atom (Cl). This molecular structure gives hypochlorous acid its unique properties, such as its ability to oxidize and disinfect.
Hypochlorous acid is a weak acid, meaning it does not fully dissociate in water. This property contributes to its stability and effectiveness in various applications. The chemical formula for hypochlorous acid is key to understanding how it interacts with other substances and why it is so effective in disinfection and sanitization.
How Does Hypochlorous Acid Work?
Hypochlorous acid works by oxidizing the cell walls of microorganisms, effectively destroying their structure and rendering them harmless. This process occurs because the chemical formula for hypochlorous acid allows it to release oxygen atoms, which are highly reactive. These oxygen atoms attack the lipids and proteins in the cell membranes of bacteria, viruses, and fungi, leading to their destruction.
Unlike other disinfectants, hypochlorous acid is gentle on human tissues, making it safe for use in wound care and skin treatments. Its ability to kill pathogens without causing harm to living tissues is one of the reasons it is widely used in healthcare settings.
Read also:Hwang In Yeop Is He Married And What We Know About His Love Life
What Are the Applications of Hypochlorous Acid?
The applications of hypochlorous acid are vast and varied. Here are some of the most common uses:
- Water Treatment: Hypochlorous acid is used to purify drinking water and treat wastewater due to its ability to kill harmful microorganisms.
- Healthcare: It is used in wound care, surface disinfection, and as a sanitizer in medical facilities.
- Agriculture: Farmers use hypochlorous acid to sanitize equipment and prevent the spread of diseases in crops and livestock.
- Household Cleaning: It is a popular ingredient in eco-friendly cleaning products due to its non-toxic nature.
Is Hypochlorous Acid Safe to Use?
Yes, hypochlorous acid is considered safe for use in various applications. Its chemical formula for hypochlorous acid ensures that it breaks down into harmless byproducts, such as water and salt, after use. This makes it an environmentally friendly option compared to other disinfectants that may leave harmful residues.
However, it is essential to use hypochlorous acid as directed, as improper use can lead to reduced effectiveness or potential harm. For example, exposure to high concentrations of hypochlorous acid can irritate the skin or respiratory system. Always follow safety guidelines when handling this compound.
How is Hypochlorous Acid Produced?
Hypochlorous acid is typically produced through the electrolysis of a saline solution. During this process, an electric current is passed through saltwater, causing the formation of hypochlorous acid. This method is widely used in industrial settings to produce large quantities of the compound for commercial use.
Another method involves dissolving chlorine gas in water, which results in the formation of hypochlorous acid. While this method is effective, it requires careful handling due to the toxic nature of chlorine gas. Regardless of the production method, the chemical formula for hypochlorous acid remains consistent, ensuring its effectiveness in various applications.
What Are the Chemical Properties of Hypochlorous Acid?
Hypochlorous acid is a weak acid with a molecular weight of approximately 52.46 g/mol. It is highly soluble in water and exists in equilibrium with its dissociated ions, hypochlorite (OCl⁻) and hydrogen (H⁺). The chemical formula for hypochlorous acid, HOCl, reflects its molecular structure, which is responsible for its oxidizing properties.
One of the key properties of hypochlorous acid is its instability. It decomposes quickly when exposed to light, heat, or organic matter, which is why it is often stored in dark, cool environments. Despite its instability, hypochlorous acid remains highly effective in killing pathogens, making it a valuable compound in various industries.
What Are the Benefits of Hypochlorous Acid?
Hypochlorous acid offers numerous benefits, making it a versatile and valuable compound. Here are some of its key advantages:
- Non-Toxic: Hypochlorous acid is safe for use on human skin and tissues, making it ideal for wound care and skincare products.
- Eco-Friendly: It breaks down into harmless byproducts, reducing its environmental impact.
- Effective Disinfectant: The chemical formula for hypochlorous acid allows it to kill a wide range of pathogens, including bacteria, viruses, and fungi.
- Versatile: It can be used in various industries, from healthcare to agriculture, due to its broad spectrum of applications.
Common Misconceptions About Hypochlorous Acid
Despite its widespread use, there are several misconceptions about hypochlorous acid. One common myth is that it is the same as bleach. While both compounds contain chlorine, they are chemically different. Bleach, or sodium hypochlorite, is a strong base, whereas hypochlorous acid is a weak acid with a different chemical formula.
Another misconception is that hypochlorous acid is harmful to humans. In reality, it is safe for use on skin and tissues when used as directed. Understanding the chemical formula for hypochlorous acid and its properties can help dispel these myths and highlight its benefits.
Frequently Asked Questions About Hypochlorous Acid
What is the chemical formula for hypochlorous acid?
The chemical formula for hypochlorous acid is HOCl. This formula represents a molecule consisting of one hydrogen atom, one oxygen atom, and one chlorine atom.
Is hypochlorous acid the same as bleach?
No, hypochlorous acid is not the same as bleach. While both compounds contain chlorine, they have different chemical formulas and properties. Bleach is a strong base, whereas hypochlorous acid is a weak acid.
How does the chemical formula for hypochlorous acid contribute to its effectiveness?
The chemical formula for hypochlorous acid, HOCl, allows it to release oxygen atoms, which are highly reactive. These oxygen atoms oxidize the cell walls of microorganisms, effectively destroying them.
What are the main uses of hypochlorous acid?
Hypochlorous acid is used in water treatment, healthcare, agriculture, and household cleaning due to its antimicrobial properties and eco-friendly nature.
In conclusion, hypochlorous acid is a remarkable compound with a wide range of applications. Its chemical formula, HOCl, is the foundation of its effectiveness as a disinfectant and sanitizer. By understanding its properties, benefits, and uses, you can appreciate why it is so highly valued in various industries. Whether you're a student, researcher, or simply curious about chemistry, this guide provides a comprehensive overview of the chemical formula for hypochlorous acid and its significance in modern science.
Hwang Dong-Hyuk Net Worth: A Deep Dive Into The Success Of South Korea's Acclaimed Filmmaker
Nahla Ariela Aubry 2024: A Glimpse Into Her Life And Journey
Discovering The World Of Kit Marc Maron: A Comprehensive Guide
HClO Hypochlorous Acid Molecule Model and Chemical Formula Stock Vector

Hypochlorous Acid Hclo Molecule Chemical Structure Stock Illustration